
The precipitate DOES exist as a "molecular" species, the water does not break the crystal lattice apart, so the silver chloride, AgCl (s), exists as a solid NOT as ions surrounded by water molecules.

Aqueous sodium nitrate solution, NaNO 3(aq), contains sodium ions, Na + (aq), and nitrate ions, NO 3 - (aq).If a salt is soluble in water, it dissolves in water by breaking up into its ions which are completely surrounded by water, so the species in water are not "salt molecules" but cations and anions surrounded by water molecules: The possible products are salts made up of a cation (positively charged ion) and an anion (negatively charged ion): Aqueous silver nitrate solution, AgNO 3(aq), contains silver ions, Ag + (aq), and nitrate ions, NO 3 - (aq).Aqueous sodium chloride solution, NaCl (aq), contains sodium ions, Na + (aq), and chloride ions, Cl - (aq).We can work out the possible products of the reaction by looking at the species present in each solution: Spectator ions are not included in the equation:Ĭonsider the reaction between aqueous solutions of sodium chloride, NaCl(aq), and silver nitrate, AgNO 3(aq). Only the reactants and product taking part in the formation of the precipitate are written in the equation, the reactants as ions, the product as a molecule. Chemical equations written to represent precipitation reactions can be written in one of three ways 3:Īll reactants and products are written as if they are molecules:Įxample: AB (aq) + CD (aq) → AD (aq) + CB (s)Īll reactants and products that are soluble are written as ions, only the precipitate is written as if it were a molecule:Įxample: A + (aq) + B - (aq) + C + (aq) + D - (aq) → A + (aq) + D - (aq) + CB (s).States of matter must be included in the chemical equation for a precipitation reaction:.
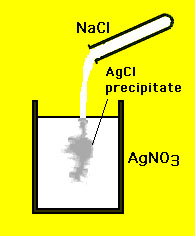
Spectator ions do not participate in precipitation, they "watch" the action like spectators watching sport. Spectator ions are ions in solution that are not used to form the precipitate.A precipitation reaction is a chemical reaction which produces a precipitate when solutions are mixed together.Solubility Rules can be used to predict if a product will be insoluble (forms a precipitate) 2 in aqueous solutions at 25☌ A precipitate is an insoluble 1 solid that forms when solutions are mixed.You need to become an AUS-e-TUTE Member! Precipitation Reaction Equations Chemistry Tutorial Key Concepts Want chemistry games, drills, tests and more? Precipitation Reaction Equations Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us
